Most people can relate to discomfort from heartburn and regurgitation, two of the most common symptoms caused by GERD. Symptoms often occur during or after eating and drinking, which affects both motivation and the ability to enjoy social activities such as picnics, dinners, events, or just a cup of coffee with friends. More severe stages of GERD with the presence of esophageal erosions (erosive GERD) or ulcerations (complicated GERD) can be very painful.
Clinical Stage Pharma
Developing small molecules for the treatment of gastric acid related diseases.
The Next Leap Forward
Blockbuster Proton Pump Inhibitors (PPIs) such as Losec and Nexium have not been able to provide all gastroesophageal reflux disease (GERD) patients with sufficient disease control. The time has now come for Potassium-Competitive Acid Blockers (PCABs) to take the next leap forward in the treatment of acid-related diseases. Cinclus Pharma’s clinical stage drug candidate linaprazan glurate takes a new approach to provide improved management of GERD and for the treatment of Helicobacter Pylori (H.Pylori) infection.

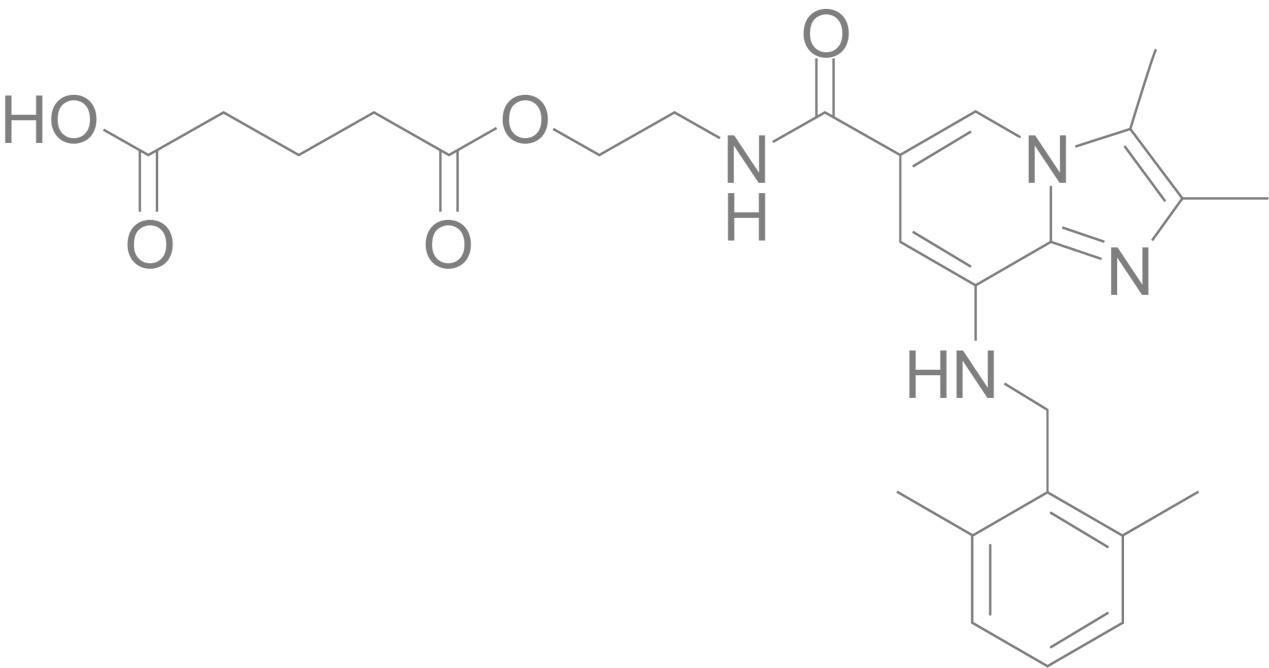
Linaprazan glurate
Built on world-leading industrial tradition, linaprazan glurate is a gastric acid blocker and therapy with great potential for healing GERD. Several clinical Phase I trials have been successfully completed and in 2022 the clinical Phase II trial was completed. Linaprazan glurate represents an improved mode of action with the potential for superior clinical efficacy and beneficial pharmacokinetic profile compared to PPIs.
In the spotlight

Cinclus Pharma is proud to be the Gold Sponsor of the OESO-SEMPIRE World Conference in Esophagology
This international congress brings together leading experts, researchers and healthcare professionals from around the world to share knowledge, discuss the latest advances in esophagology, and shape the future of esophageal care and treatment.

Cinclus Pharma participates in Digestive Disease Week (DDW) in Chicago May 2-5 2026
DDW brings together experts from across the globe for a comprehensive program of scientific sessions and networking opportunities.

CEO Christer Alhberg presents at Redeye Healthcare Day 2026
Watch the full presentation by CEO Christer Ahlberg about Cinclus Pharma and the development of linaprazan glurate, presented at Redeye Healthcare Day 2026.

CHRISTER AHLBERG
CEO CINCLUS PHARMA



